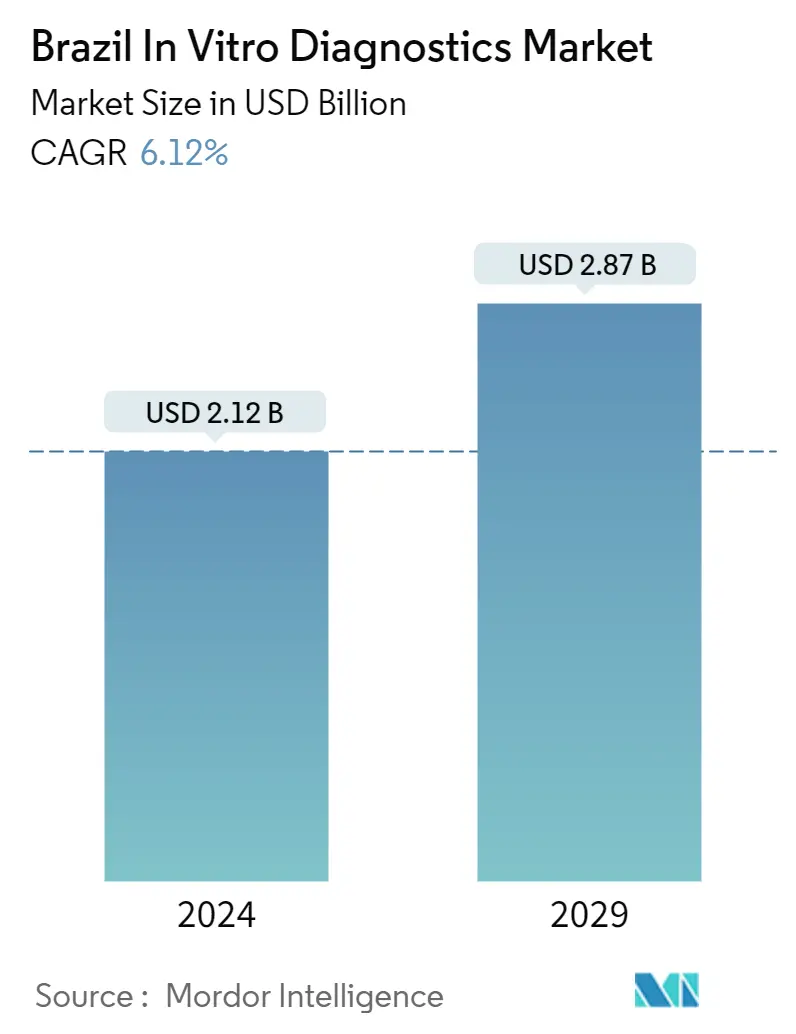
Brazil In-Vitro Diagnostics Market Size
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| Forecast Data Period | 2024 - 2029 |
| Market Size (2024) | USD 2.12 Billion |
| Market Size (2029) | USD 2.87 Billion |
| CAGR (2024 - 2029) | 6.12 % |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Brazil In-Vitro Diagnostics Market Analysis
The Brazil In Vitro Diagnostics Market size is estimated at USD 2.12 billion in 2024, and is expected to reach USD 2.87 billion by 2029, growing at a CAGR of 6.12% during the forecast period (2024-2029).
The COVID-19 pandemic significantly impacted Brazil in-vitro diagnostics market. For instance, as per a May 2020 update, Brazil's National Health Surveillance Agency (ANVISA) expedited emergency reviews and approvals for medical devices and in-vitro diagnostics (IVDs) related to the COVID-19 pandemic. The agency allowed the emergency use of information from foreign regulatory authorities that participate in the Medical Device Single Audit Program (MDSAP) in place of its own good manufacturing practice inspections.
The major factors fueling the market growth are the high prevalence of chronic diseases and the increasing use of point-of-care diagnostics.
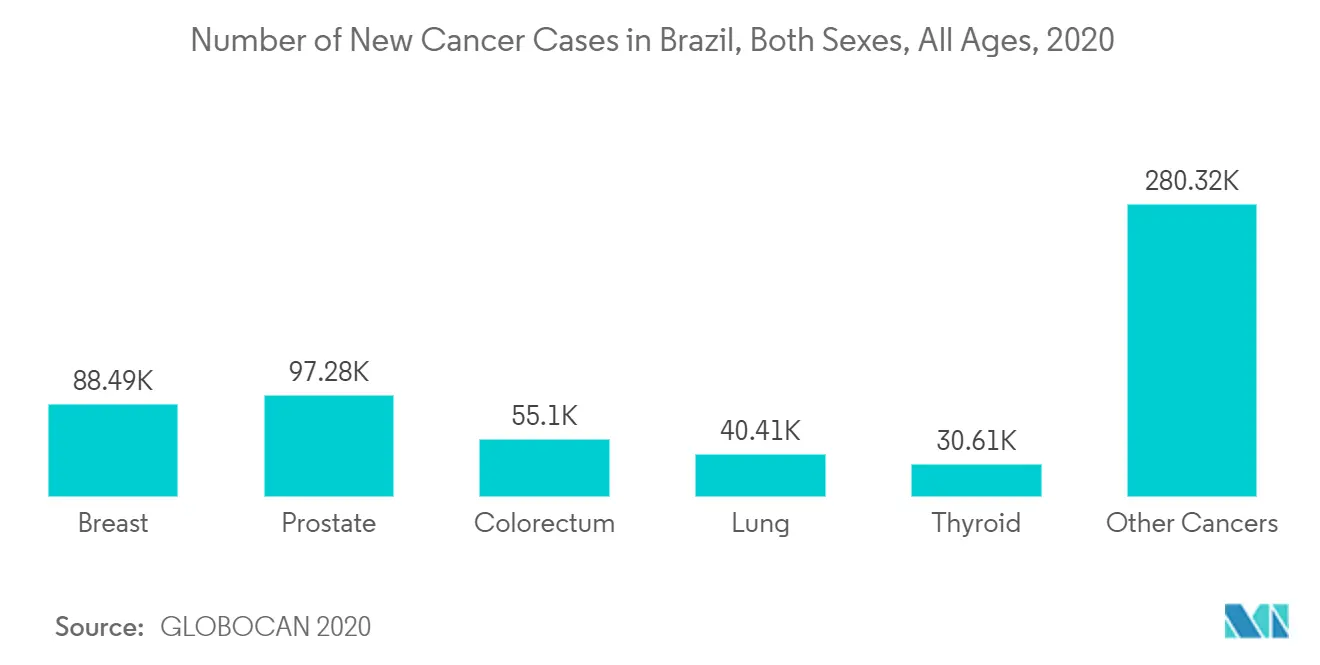
The high prevalence of chronic diseases is a major factor driving the market growth. According to World Population Ageing 2019, the number of people aged above 65 years in 2019 in Brazil region are 19.526 million and is expected to reach 52.026 million by 2050. This increase in geriatric population is expected to increase the prevalence rate of liver cancer in the country as elderly people are more prone to chronic diseases, thus boosting the market. Additionaly, leukemia is the most common cancer in children and has the incidence rates in industrialized countries, followed by developing countries. As per the GLOBOCAN 2020, 11,396 cases of leukemia were identified in Brazil in 2020 including both males and females of all ages. Such a prevalence indicates the rising demand for acute myeloid leukemia treatment in the country.
However, stringent regulations is a major factor hindering the Brazil in-vitro diagnostics market growth.
Brazil In-Vitro Diagnostics Market Trends
This section covers the major market trends shaping the Brazil In Vitro Diagnostics Market according to our research experts:
Molecular Diagnostics Segment is Expected to Witness a Significant Growth
By test type, molecular diagnostics are expected to witness significant growth. Molecular diagnostic devices are used to analyze biological markers in the genome and proteome to detect pathogens or mutations.
Factors, such as large outbreaks of bacterial and viral epidemics in the world, increasing demand for point-of-care diagnostics, and rapidly evolving technology, are driving the growth of the molecular diagnostics segment. For instance, in November 2020, Chembio Diagnostics, Inc. a leading point-of-care diagnostic company focused on infectious diseases, revealed that its subsidiary Chembio Diagnostics Brazil Ltda. received Agência Nacional de Vigilância Sanitária (ANVISA) approval for the DPP SARS-CoV-2 Antigen test system in Brazil.
The use of molecular diagnostic techniques in the development and subsequent administration of personalized medicine is likely to boost the market growth. For instance, in January 2022, the Oswaldo Cruz Foundation finished the development of two new molecular tests for the diagnosis of COVID-19, including one test submitted to ANVISA.
Thus, due to the above-mentioned factors, the market is expected to witness a significant growth over the forecast period.
Brazil In-Vitro Diagnostics Industry Overview
Danaher Corporation, Roche Diagnostics, Becton, Dickinson and Company, �����Dz�é������ܳ�, and Bio-Rad Laboratories Inc. are some of the major players in the market studied. The major companies are focusing on increasing technological advancements, in order to address customer needs, like fully automated systems, which reduces manual processes and consumables and also provides fast and accurate results.
Brazil In-Vitro Diagnostics Market Leaders
-
�����Dz�é������ܳ�
-
Danaher Corporation
-
Becton, Dickinson and Company
-
Bio-Rad Laboratories Inc.
-
Roche Diagnostics
*Disclaimer: Major Players sorted in no particular order
Brazil In-Vitro Diagnostics Market News
- In December 2020, HalioDx SAS, the immuno-oncology diagnostic company pioneering the immunological diagnosis of cancers, entered a distribution agreement with Diagnosticos da America (DASA) SA for Immunoscore commercialization in Brazil.
- In July 2020, Todos Medical Ltd. an in-vitro diagnostics company revealed that its partner supplier 3D Medicines Corporation (3D Med) has received authorization from the Brazilian Health Surveillance Agency (ANVISA) to distribute its COVID-19 qPCR test kits and ANDis extraction systems.
Brazil In-Vitro Diagnostics Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Deliverables
1.2 Study Assumptions
1.3 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 High Prevalence of Chronic Diseases
4.2.2 Increasing Use of Point-of-care (POC) Diagnostics
4.3 Market Restraints
4.3.1 Stringent Regulations
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value -USD Million)
5.1 By Test Type
5.1.1 Clinical Chemistry
5.1.2 Molecular Diagnostics
5.1.3 Immunodiagnostics
5.1.4 Hematology
5.1.5 Other Test Types
5.2 By Product
5.2.1 Instrument
5.2.2 Reagent
5.2.3 Other Products
5.3 By Usability
5.3.1 Disposable IVD Devices
5.3.2 Reusable IVD Devices
5.4 By Application
5.4.1 Infectious Disease
5.4.2 Diabetes
5.4.3 Cancer/Oncology
5.4.4 Cardiology
5.4.5 Autoimmune Disease
5.4.6 Nephrology
5.4.7 Other Applications
5.5 By End Users
5.5.1 Diagnostic Laboratories
5.5.2 Hospitals and Clinics
5.5.3 Other End Users
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Abbott Laboratories
6.1.2 Arkray Inc.
6.1.3 Becton, Dickinson and Company
6.1.4 bioMerieux
6.1.5 Bio-Rad Laboratories Inc.
6.1.6 Danaher Corporation
6.1.7 QIAGEN
6.1.8 Roche Diagnostics
6.1.9 Siemens AG
6.1.10 Thermo Fisher Scientific
6.1.11 Diagnosticos da America SA
6.1.12 Fujifilm Holdings Corporation
6.1.13 Wama Diagnóstica
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Brazil In-Vitro Diagnostics Industry Segmentation
As per the scope of the report, in vitro diagnostics involve medical devices and consumables that are utilized to perform in vitro tests on various biological samples. They are used for the diagnosis of various medical conditions, such as chronic diseases. In the report, a detailed analysis of the in vitro diagnostics market is presented, with specific attention toward diabetes and thalassemia. Brazil in vitro diagnostics market is segmented by test type (clinical chemistry, molecular diagnostics, immunodiagnostics, hematology, and other test types), product (instrument, reagent, and other products), usability (disposable IVD devices and reusable IVD devices), application (infectious disease, diabetes, cancer/oncology, cardiology, autoimmune disease, nephrology, and other applications), and end user (diagnostic laboratories, hospitals and clinics, and other end users). The report offers the value (in USD million) for the above segments.
| By Test Type | |
| Clinical Chemistry | |
| Molecular Diagnostics | |
| Immunodiagnostics | |
| Hematology | |
| Other Test Types |
| By Product | |
| Instrument | |
| Reagent | |
| Other Products |
| By Usability | |
| Disposable IVD Devices | |
| Reusable IVD Devices |
| By Application | |
| Infectious Disease | |
| Diabetes | |
| Cancer/Oncology | |
| Cardiology | |
| Autoimmune Disease | |
| Nephrology | |
| Other Applications |
| By End Users | |
| Diagnostic Laboratories | |
| Hospitals and Clinics | |
| Other End Users |
Brazil In-Vitro Diagnostics Market Research FAQs
How big is the Brazil In Vitro Diagnostics Market?
The Brazil In Vitro Diagnostics Market size is expected to reach USD 2.12 billion in 2024 and grow at a CAGR of 6.12% to reach USD 2.87 billion by 2029.
What is the current Brazil In Vitro Diagnostics Market size?
In 2024, the Brazil In Vitro Diagnostics Market size is expected to reach USD 2.12 billion.
Who are the key players in Brazil In Vitro Diagnostics Market?
�����Dz�é������ܳ�, Danaher Corporation, Becton, Dickinson and Company, Bio-Rad Laboratories Inc. and Roche Diagnostics are the major companies operating in the Brazil In Vitro Diagnostics Market.
What years does this Brazil In Vitro Diagnostics Market cover, and what was the market size in 2023?
In 2023, the Brazil In Vitro Diagnostics Market size was estimated at USD 2 billion. The report covers the Brazil In Vitro Diagnostics Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Brazil In Vitro Diagnostics Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Brazil In-Vitro Diagnostics Industry Report
The Brazil In-Vitro Diagnostics market is comprehensively analyzed in this report, which segments the market by test type, product, usability, application, and end user. The test types covered include clinical chemistry, molecular diagnostics, immunodiagnostics, hematology, and other test types. The product segmentation includes instruments, reagents, and other products. The usability segment is divided into disposable IVD devices and reusable IVD devices. Applications encompass infectious disease, diabetes, cancer/oncology, cardiology, autoimmune disease, nephrology, and other applications. End users include diagnostic laboratories, hospitals and clinics, and other end users.
This market report delves into the market size, market trends, and market share, providing a detailed market analysis and market forecast. The industry trends and industry report offer insights into the growth rate and industry outlook. The report includes industry statistics and industry research, giving a comprehensive view of the market data and market growth.
The industry information provided in this report includes a market overview and market segmentation, highlighting the market leaders and market value. The report example and report pdf offer a clear representation of the market predictions and market review. Research companies can benefit from the industry sales and market outlook provided in this report, which includes market predictions and market review.
Overall, this report provides a thorough industry analysis and market forecast, offering valuable insights into the Brazil In-Vitro Diagnostics market.



