Duchenne Muscular Dystrophy Treatment Market Size
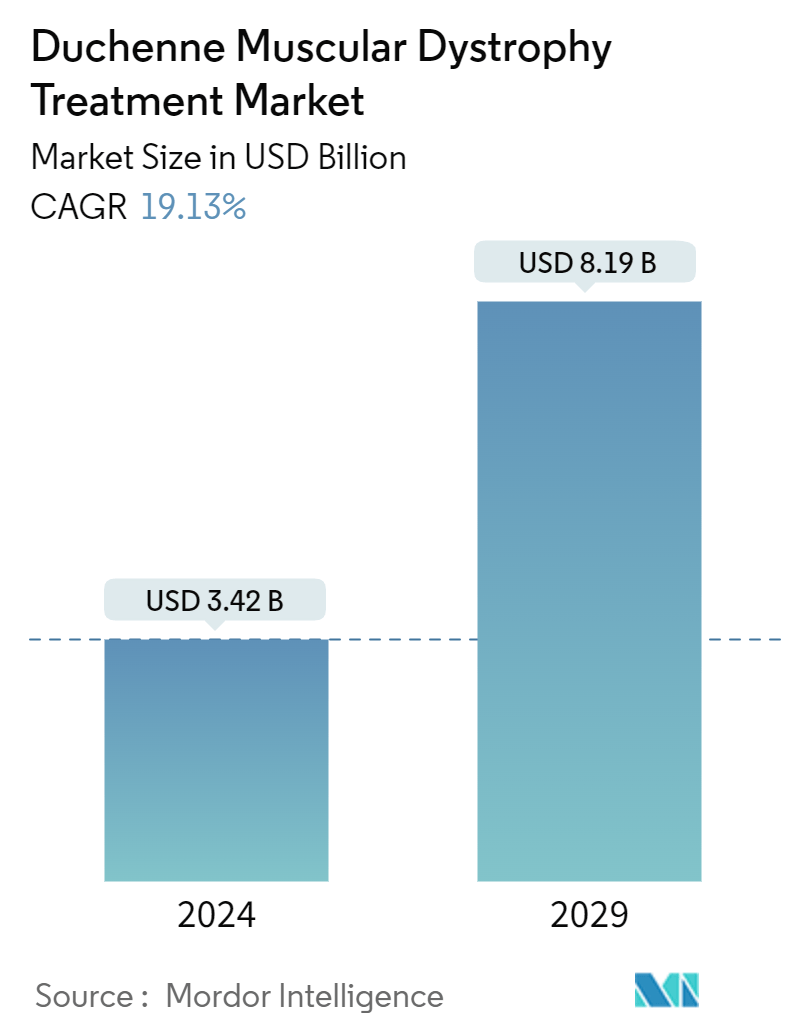
| Study Period | 2021 - 2029 |
| Market Size (2024) | USD 3.42 Billion |
| Market Size (2029) | USD 8.19 Billion |
| CAGR (2024 - 2029) | 19.13 % |
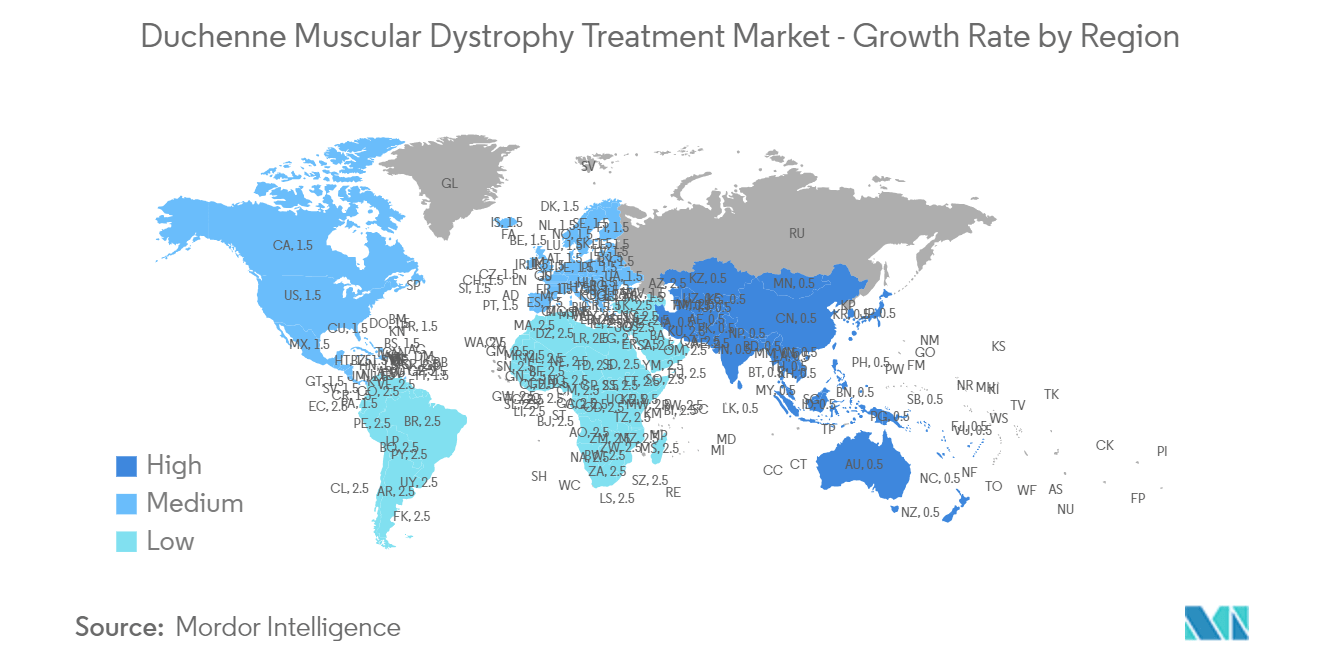
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
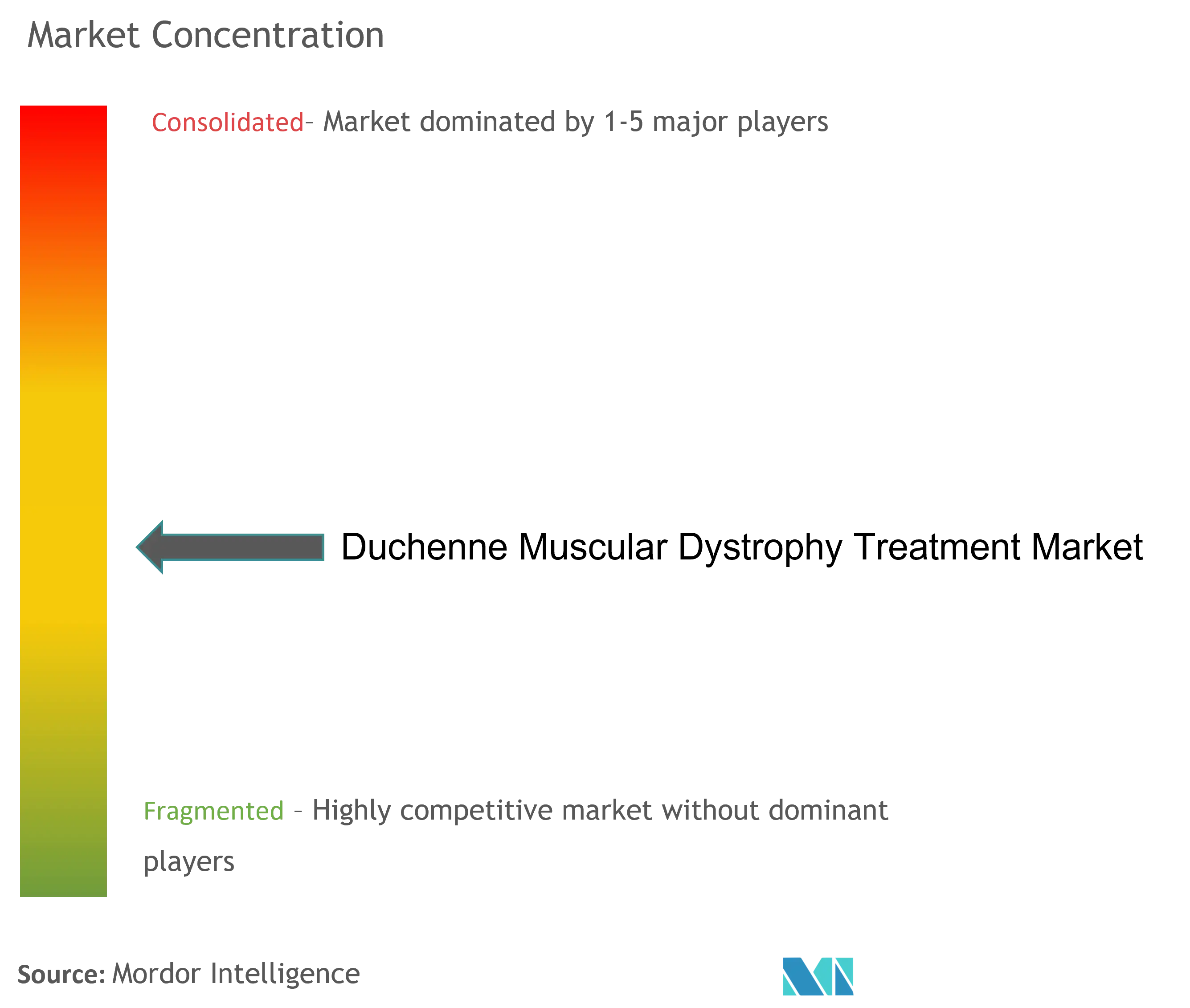
| Market Concentration | Medium |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Duchenne Muscular Dystrophy Treatment Market Analysis
The Duchenne Muscular Dystrophy Treatment Market size is estimated at USD 3.42 billion in 2024, and is expected to reach USD 8.19 billion by 2029, growing at a CAGR of 19.13% during the forecast period (2024-2029).
The rising burden of Duchenne muscular dystrophy (DMD), increasing investments in the R&D of novel therapies for DMD, and increasing awareness campaigns for DMD are the major drivers for the market. For instance, according to the data published by Muscular Dystrophy Association Inc. in June 2023, the prevalence of DMD in Europe and North America is approximately 6 per 100,000 individuals. According to a study published in the PLOS One Journal in June 2023, the prevalence of DMD within the general populace ranges from 1.7 to 3.4 cases per 100,000 individuals. Additionally, the birth prevalence of DMD stands at 21.7-28.2 cases per 100,000 live male births. These statistics provide valuable insights for healthcare professionals and stakeholders involved in managing and treating DMD, facilitating a better understanding of the condition's impact and informing strategic decision-making in healthcare provision and resource allocation. Thus, the high burden of this rare genetic disease is expected to boost the demand for its effective therapy, thereby boosting market growth.
Increasing investments in R&D by the private and public sectors have also increased the number of clinical trials evaluating potential DMD treatments. The approved pharmacological therapies for managing DMD are based on exon skipping therapies, mutation suppressing therapies, gene therapies, and corticosteroids, among others. In recent years, there has been a significant increase in the discovery and development of pharmaceutical drugs. Nearly all major companies are now focusing on R&D, which is likely to impact the market significantly in the coming years. For instance, in January 2023, Parent Project Muscular Dystrophy (PPMD) disclosed a strategic investment of USD 500,000 in Myosana Therapeutics Inc. (Myosana) to bolster the advancement and implementation of its non-viral gene therapy delivery platform. This initiative is geared toward mitigating skeletal muscle degeneration and addressing heart failure associated with DMD. Thus, increasing cases of DMD and rising investments in research and development are expected to propel the demand for the treatment, which may positively impact the market's growth.
Mutation-specific therapies may become a breakthrough in treating DMD. Additionally, there has been an increase in the number of campaigns to raise awareness of DMD, the development of mutation-specific therapies, the prevalence of the disease, the number of products in development, and the introduction of therapies like Exondys51 and Translarna, as well as corticosteroid-based anti-inflammatory drugs. The advancement of innovative pharmaceuticals and therapies, including disease-modifying treatments, substantial investment from multiple firms in drug research, and advocacy efforts from patient groups influencing regulatory approval processes, are primary catalysts propelling the Duchenne muscular dystrophy treatment market.
However, the lack of standardization to measure clinical efficacy across all stages of DMD may hinder the market’s growth over the forecast period.
Duchenne Muscular Dystrophy Treatment Market Trends
The Exon-skipping Segment is Expected to Hold a Major Market Share Over the Forecast Period
Mutations involving internal deletions in the gene responsible for encoding dystrophin, a protein vital for maintaining muscle cell membrane integrity, are the predominant drivers of Duchenne muscular dystrophy. A promising therapeutic avenue involves the strategic masking of an exon located proximal to the deleted regions, facilitating the alignment of the remaining exons. This approach, known as exon-skipping, is one of the most efficacious treatment modalities for reinstating the production of a truncated yet functional dystrophin protein.
Market players focus on frequent launches in exon skipping technology, a significant factor driving the segment’s growth. For instance, in November 2023, six therapies were approved in Europe for treating DMD, including four exon-skipping drugs (eteplirsen, viltolarsen, golodiresen, and casimersen). These drugs require lifetime treatment and work to trick muscle cells into generating a slightly shorter but functional dystrophin protein. This approach bypasses the effect of the disease-causing mutation. Similarly, in January 2024, Sarepta Therapeutics Inc. reported positive data from Part B of the MOMENTUM study (Study SRP-5051-201), a global, Phase 2, multi-ascending dose clinical trial of SRP-5051 (vesleteplirsen) that enrolled patients aged 8-21 years. SRP-5051 represents a cutting-edge peptide phosphorodiamidate morpholino oligomer (PPMO) therapy designed for individuals afflicted with DMD who exhibit exon 51 susceptibility. Consequently, substantial segment expansion is anticipated throughout the projected period, owing to product introductions, regulatory endorsements, and technological innovations.
North America is Expected to Witness Considerable Growth Over the Forecast Period
North America is expected to grow considerably due to new product innovations, high healthcare expenditure, and government awareness programs. Due to the increased disease prevalence and anticipated launches of attractive pipeline candidates, the United States has led the regional market and is expected to keep this position. The market is poised for growth with the increasing number of clinical trials conducted worldwide, notably within the United States. For instance, in June 2023, Sarepta Therapeutics Inc. obtained accelerated approval from the US Food and Drug Administration (FDA) for ELEVIDYS (delandistrogene moxeparvovec-rokl), an adeno-associated virus-based gene therapy. This therapy targets the treatment of ambulatory pediatric patients aged 4-5 years diagnosed with DMD and confirmed mutations in the DMD gene. Similarly, in March 2024, Catalyst Pharmaceuticals Inc. announced the US commercial launch of AGAMREE (vamorolone) oral suspension 40 mg/mL for treating Duchenne muscular dystrophy (DMD) in patients aged two years and older.
The Duchenne muscular dystrophy treatment market is expected to grow significantly in the region due to huge unmet needs and favorable initiatives by key players. For instance, in May 2023, PepGen Inc. received a No Objection Letter (NOL) for its Clinical Trial Application (CTA) from Health Canada for its Phase 2 CONNECT1-EDO51 study to initiate an open-label, multiple ascending dose (MAD) clinical trial of PGN-EDO51 in patients with Duchenne muscular dystrophy (DMD) amenable to an exon 51 skipping approach. The presence of several new medications in the pipeline, the availability of gene therapies, and the anticipated increase in treatment rates will be significant growth drivers for the DMD market. Thus, significant market growth is expected over the forecast period in North America.
Duchenne Muscular Dystrophy Treatment Industry Overview
The Duchenne muscular dystrophy treatment market is moderately fragmented and consists of several major players. Some companies currently dominating the market are Nippon Shinyaku Co. Ltd, ITALFARMACO SpA, PTC Therapeutics, Santhera Pharmaceuticals Holding AG, and Sarepta Therapeutics Inc.
Duchenne Muscular Dystrophy Treatment Market Leaders
-
NIPPON SHINYAKU CO., LTD. (NS Pharma Inc.)
-
ITALFARMACO S.p.A.
-
PTC Therapeutics
-
Santhera Pharmaceuticals
-
Sarepta Therapeutics
*Disclaimer: Major Players sorted in no particular order
Duchenne Muscular Dystrophy Treatment Market News
- March 2024: Santhera Pharmaceuticals launched AGAMREE (vamorolone) for treating DMD in the United States. The drug was launched by Catalyst Pharmaceuticals Inc., Santhera's commercialization partner for North America.
- February 2024: BioMarin conducted Phase 1/2, an open-label, multi-center study consisting of two parts to evaluate the safety and tolerability of BMN 351 at escalating doses in participants with DMD, with genetic mutations amenable to exon 51 skipping.
Duchenne Muscular Dystrophy TreatmentMarket Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Rising Disease Burden of Duchenne Muscular Dystrophy (DMD)
4.2.2 Increasing Investments in the R&D of Novel Therapies for DMD
4.2.3 Increasing Awareness Campaigns for DMD
4.3 Market Restraints
4.3.1 Lack of Standardization to Measure Clinical Efficacy Across All Stages of DMD
4.4 Porter's Five Forces Analysis
4.4.1 Bargaining Power of Buyers/Consumers
4.4.2 Bargaining Power of Suppliers
4.4.3 Threat of New Entrants
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value – USD)
5.1 By Therapeutic Approaches
5.1.1 Molecular-based Therapies
5.1.1.1 Mutation Suppression
5.1.1.2 Exon Skipping
5.1.2 Steroid Therapy
5.1.3 Other Therapeutic Approaches
5.2 Geography
5.2.1 North America
5.2.1.1 United States
5.2.1.2 Canada
5.2.1.3 Mexico
5.2.2 Europe
5.2.2.1 Germany
5.2.2.2 United Kingdom
5.2.2.3 France
5.2.2.4 Italy
5.2.2.5 Spain
5.2.2.6 Rest of Europe
5.2.3 Asia-Pacific
5.2.4 Rest of the World
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 NIPPON SHINYAKU CO. LTD (NS Pharma Inc.)
6.1.2 ITALFARMACO SpA
6.1.3 PTC Therapeutics
6.1.4 Santhera Pharmaceuticals
6.1.5 Sarepta Therapeutics
6.1.6 Fibrogen Inc.
6.1.7 BioMarin
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Duchenne Muscular Dystrophy Treatment Industry Segmentation
As per the scope of this report, Duchenne muscular dystrophy is a rare genetic disease that causes gradual muscle wasting and weakness, brought on by the X-linked recessive pattern, which causes muscle deterioration. However, it could also be a recent mutation or a parent-passed genetic characteristic. The Duchenne muscular dystrophy treatment market is segmented by therapeutic approaches and geography. By therapeutic approaches, the market is further segmented into molecule-based therapies, steroid therapy, and other therapeutic approaches. The report also covers the market sizes and forecasts for major countries across different regions. The market size is provided for each segment in terms of value (USD).
| By Therapeutic Approaches | ||||
| ||||
| Steroid Therapy | ||||
| Other Therapeutic Approaches |
| Geography | ||||||||
| ||||||||
| ||||||||
| Asia-Pacific | ||||||||
| Rest of the World |
Duchenne Muscular Dystrophy TreatmentMarket Research FAQs
How big is the Duchenne Muscular Dystrophy Treatment Market?
The Duchenne Muscular Dystrophy Treatment Market size is expected to reach USD 3.42 billion in 2024 and grow at a CAGR of 19.13% to reach USD 8.19 billion by 2029.
What is the current Duchenne Muscular Dystrophy Treatment Market size?
In 2024, the Duchenne Muscular Dystrophy Treatment Market size is expected to reach USD 3.42 billion.
Who are the key players in Duchenne Muscular Dystrophy Treatment Market?
NIPPON SHINYAKU CO., LTD. (NS Pharma Inc.), ITALFARMACO S.p.A., PTC Therapeutics, Santhera Pharmaceuticals and Sarepta Therapeutics are the major companies operating in the Duchenne Muscular Dystrophy Treatment Market.
Which is the fastest growing region in Duchenne Muscular Dystrophy Treatment Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Duchenne Muscular Dystrophy Treatment Market?
In 2024, the North America accounts for the largest market share in Duchenne Muscular Dystrophy Treatment Market.
What years does this Duchenne Muscular Dystrophy Treatment Market cover, and what was the market size in 2023?
In 2023, the Duchenne Muscular Dystrophy Treatment Market size was estimated at USD 2.77 billion. The report covers the Duchenne Muscular Dystrophy Treatment Market historical market size for years: 2021, 2022 and 2023. The report also forecasts the Duchenne Muscular Dystrophy Treatment Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Duchenne Muscular Dystrophy TreatmentIndustry Report
The Duchenne Muscular Dystrophy Treatment market is segmented by therapeutic approaches, including molecule-based therapies, steroid therapy, and other therapeutic approaches. The geographical segmentation covers North America, Europe, Asia-Pacific, and the rest of the world. This industry analysis provides a comprehensive market overview, focusing on market size and market share within these segments.
The market research highlights key market trends and market growth, supported by industry reports and industry statistics. The market forecast offers insights into the market outlook and market predictions for the coming years. The report also delves into the market segmentation, providing a detailed market review and market value assessment.
The industry research includes an industry overview, showcasing the industry outlook and industry trends. Market leaders are identified, and their contributions to market growth are discussed. The report example and report PDF download offer additional industry information and industry sales data.
This market analysis emphasizes the importance of understanding market data and market forecast to predict future market growth. The industry analysis includes a historical overview and a market forecast outlook. Overall, the report provides a thorough examination of the Duchenne Muscular Dystrophy Treatment market, ensuring a well-rounded understanding of its current state and future potential.



