Market Trends of Global Hereditary Angioedema Therapeutics Industry
This section covers the major market trends shaping the Hereditary Angioedema Therapeutics Market according to our research experts:
The Kallikrein Inhibitors Segment is the Fastest Growing in the Hereditary Angioedema Therapeutics Market
Kallikrein inhibitors are expected to be the fastest-growing segment over the forecast period. Currently, various kallikrein inhibitors are being evaluated due to their potential to prevent edematous HAE attacks. This comparatively emerging class of drug therapy has several potential molecules in the pipelines, the commercialization of which is expected to fuel the market growth. For instance, BioCryst is developing a novel treatment to treat and prevent angioedema attacks in patients with hereditary angioedema (HAE) and evaluating the molecule APeX-1, an oral, once-daily, selective inhibitor of plasma kallikrein. Also, pharmaceutical companies are rapidly seeking approval in this class of drugs due to lesser competition in this market space. For instance, in August 2020, the US Food and Drug Administration approved Takhzyro (Lanadelumab). It is the first monoclonal antibody that targets kallikrein and is indicated for treating patients aged 12 years and older with types I and II HAE.
In December 2021, BioCryst Pharmaceuticals was granted USD 350 million to help fund the global launch of Orladeyo (Berotralstat), an oral treatment for preventing hereditary angioedema (HAE) swelling attacks. It works by inhibiting kallikrein activity.
Due to such developments, the kallikrein inhibitors segment is expected to augment the market's growth. The increasing R&D and funding for orphan drug development and rare genetic disorders are expected to further fuel the market's growth. For instance, the Indian Ministry of Health and Family Welfare developed a National Policy to treat rare diseases in India to progressively expand India's capabilities to treat rare diseases. Hence, the increased funding for R&D for new drugs is propelling the segment's growth.
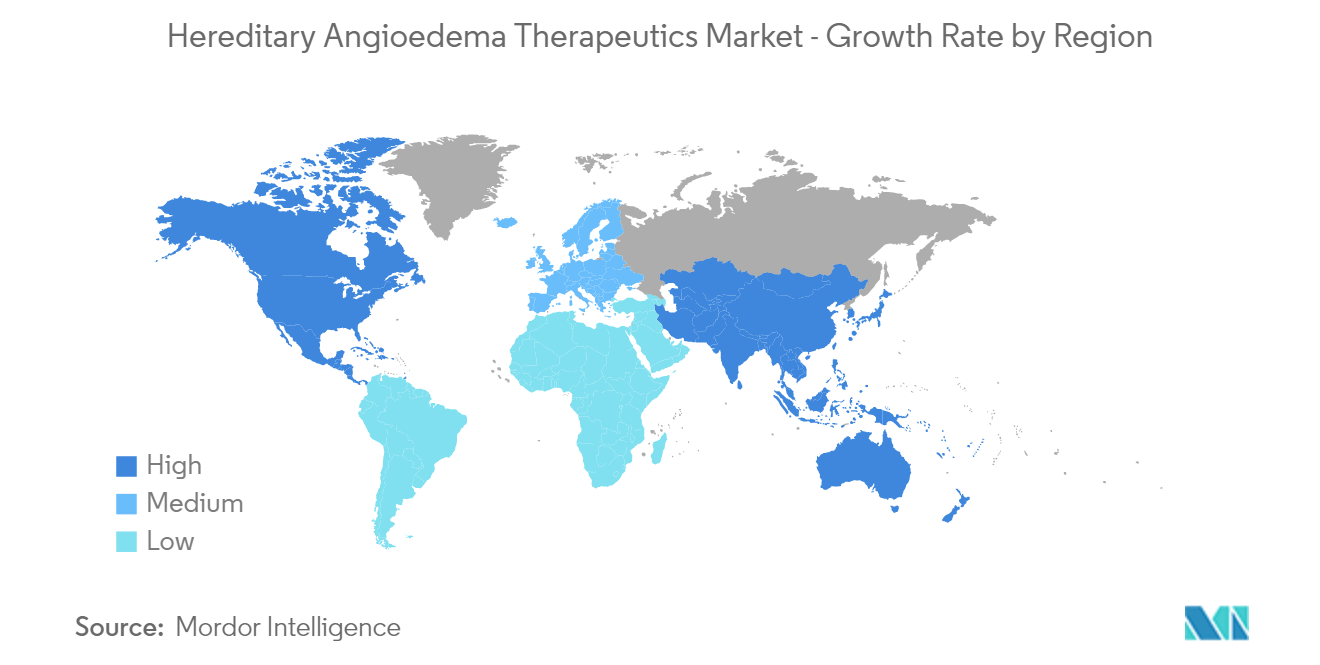
North America Dominates the Market and is Expected to Continue the Domination During the Forecast Period
North America is expected to dominate the overall hereditary angioedema market during the forecast period due to the presence of key players, increasing awareness programs and research organizations regarding the treatment of the condition, and established healthcare infrastructure. The increasing government initiatives, rising research partnerships, and a robust pipeline are the major growth drivers of the market.
For instance, Ionis Pharmaceuticals, a biotechnology company based in California, has IONIS-PKK-LRx, a ligand-conjugated (LICA) investigational antisense medicine in its pipeline. The company is evaluating the drug molecule to reduce the production of prekallikrein, or PKK, to treat patients with hereditary angioedema. In September 2021, Cycle Pharmaceuticals launched SAJAZIR (Icatibant) Injection, a new treatment option for patients affected by hereditary angioedema (HAE) approved by the US FDA. The region has many government- and non-government-funded research institutions. For instance, the National Center for Advancing Translational Sciences (NCATS) is entirely focused on esoteric testing for the cure and treatments for rare diseases. NCATS aims to get collaborative projects to study common themes and causes of related diseases to speed the development of treatments. Hence, due to such factors, the market is anticipated to have lucrative growth in the future.