Rapid Plasma Reagin Test Market Size
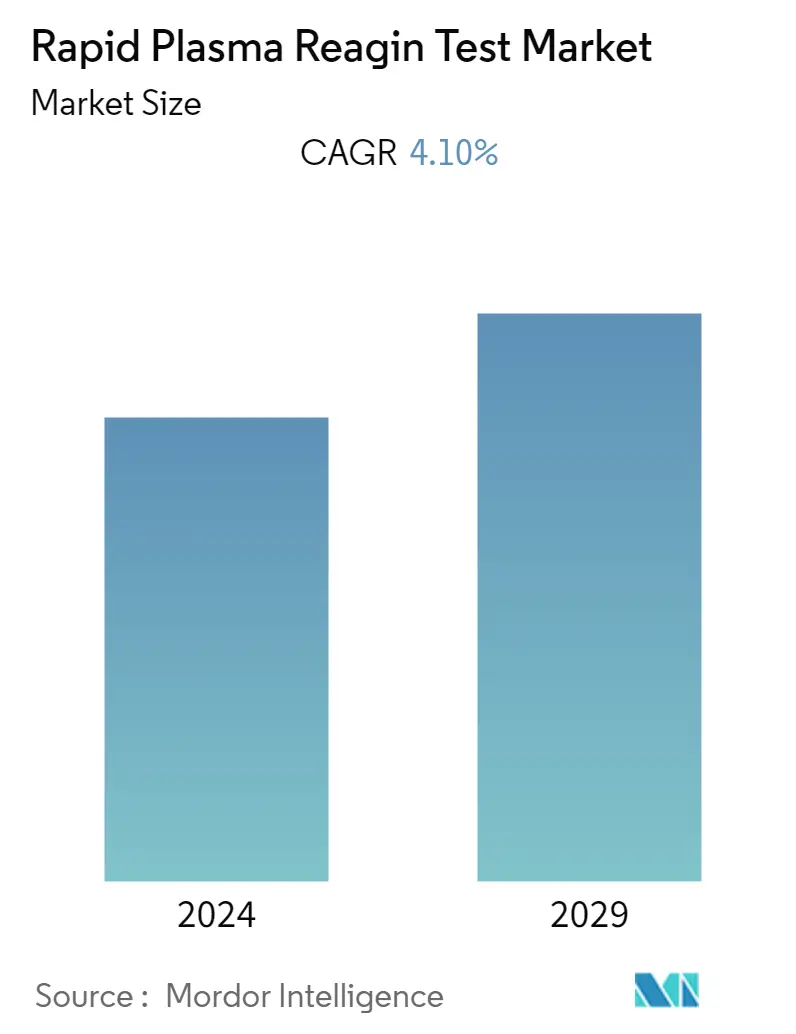
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| Forecast Data Period | 2024 - 2029 |
| CAGR | 4.10 % |
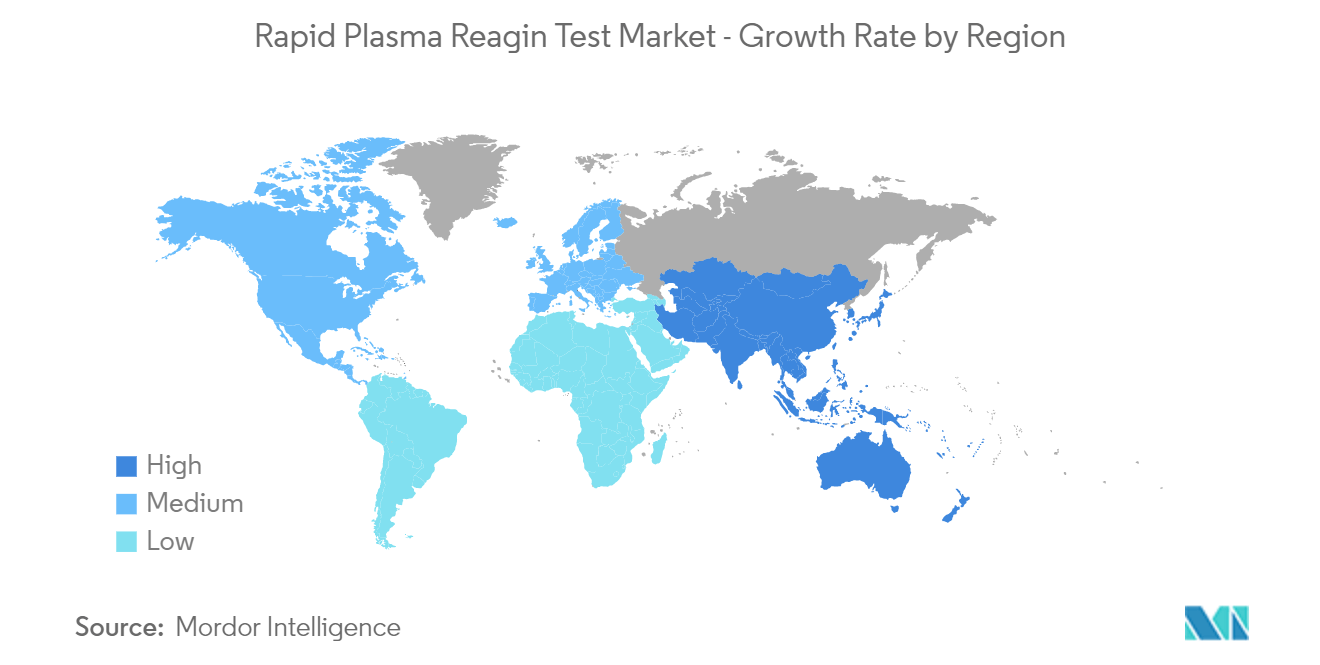
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Rapid Plasma Reagin Test Market Analysis
The market for the rapid plasma reagin test is expected to exhibit a CAGR of 4.1% during the forecast period.
- The COVID-19 pandemic had a significant impact on market growth, as syphilis diagnoses decreased during the early stages of the pandemic. However, there was an increase in syphilis infections during the post-pandemic market scenario, which drove the demand for the rapid plasma reagin test. This rise in syphilis is expected to contribute to the growth of the market during the post-pandemic period.
- The rapid plasma reagin (RPR) test is a serological test used to screen for syphilis, a sexually transmitted infection caused by the spirochete bacterium Treponema pallidum. The market for the rapid plasma reagin test is being driven by an increasing prevalence of syphilis and increased awareness of sexually transmitted diseases.
- According to a report published by the World Health Organization (WHO) in July 2021, the global pooled prevalence of syphilis among homosexuals was 7.5%, and globally, there were an estimated 7 million new syphilis infections in 2021. The high rate of syphilis prevalence is expected to drive the demand for rapid plasma reagin tests, thereby contributing to the growth of the market. Additionally, the prevalence of syphilis infections among pregnant women is expected to drive the growth of the market owing to the growing demand for early diagnosis to obtain proper treatment.
- Several initiatives are being taken regarding STD diagnostics, further supporting the growth of the market over the forecast period. For instance, in December 2021, Clinton Health Access Initiative (CHAI), MedAccess, and SD Biosensor partnered to develop dual Syphilis rapid diagnostics tests for a cost under USD 1, which is recommended by the WHO. Such initiatives are augmenting the demand for rapid tests for the rapid plasma reagin test, which is expected to contribute to the growth of the market over the forecast period. However, the lack of adoption of disease screening in developing countries is expected to restrain the growth of the market over the forecast period.
Rapid Plasma Reagin Test Market Trends
The Rapid Plasma Reagin Test Kits Segment is Expected to Hold a Significant Share in the Market Over the Forecast Period
- The rapid plasma reagin (RPR) test kits are expected to hold a significant share in the market over the forecast period due to their widespread usage for qualitative and semi-quantitative determination of reagin antibodies in serum or plasma from individuals with syphilis. The segment is anticipated to exhibit a similar trend over the forecast period because of the high prevalence of syphilis among the global population and the growing availability of test kits.
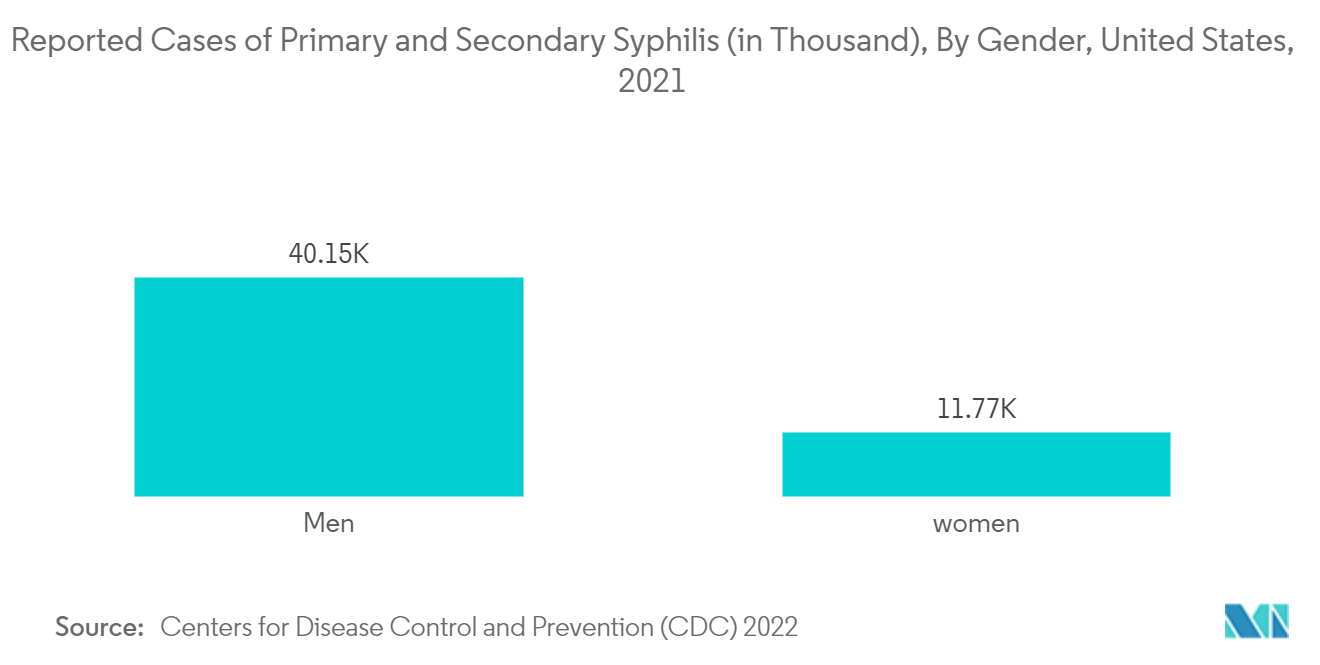
- According to the Center for Disease Control and Prevention (CDC) data published in July 2022, there were 52,354 reported cases of primary and secondary syphilis in the United States in 2021. The same source also reported 2,677 cases of congenital syphilis in the United States in 2021. Additionally, according to the World Health Organization's (WHO) 2021 data, there are 988,000 new maternal syphilis infections worldwide each year. The high incidence of syphilis among the global population is expected to drive the demand for RPR test kits, thereby contributing to the growth of the studied segment.
- As part of antenatal care, RPR tests can be utilized as the initial test for pregnant women. The increasing incidence of syphilis and technological advancements in RPR test kits are the key driving factors in the RPR test kits segment.
North America is Expected to Hold a Significant Share in the Market During the Forecast Period
- North America is anticipated to dominate the global rapid plasma reagin test market owing to the rising incidence of sexually transmitted diseases, including syphilis, and the growing awareness regarding the morbidity of such diseases in this region. For example, according to the Center for Disease Control and Prevention (CDC) data in 2022, there were 50,592 cases of early non-primary and non-secondary syphilis in the United States in 2021. As per the same source, the rate of primary and secondary syphilis per 100,000 in the United States was 15.8 in 2021.
- The high expenditure on research and development (R&D) by government organizations and pharmaceutical companies for early diagnosis and treatment is among the primary factors propelling the regional market. For instance, as per the National Centre for Health Statistics (NCHS), investments for sexually transmitted infection research in the United States in 2021 amounted to USD 404 million. Similarly, the increase in syphilis cases in Canada is projected to drive the demand for rapid plasma reagin tests, thereby contributing to the market's growth over the forecast period. For example, according to data from the Public Health Agency of Canada published in November 2021, the national rate of infectious syphilis increased from 24 per 100,000 population and rose by 124% compared to the previous years. Such a significant increase in syphilis infections is expected to boost the market growth over the forecast period.
- Likewise, the growth of the rapid plasma reagin test market is attributed to the incidence of syphilis among the Mexican population. For instance, as per the BMC article published in January 2021, the incidence of syphilis increased by an average of 0.336 cases per 100,000 per year and was higher among women aged 15-19 years in Mexico. Such a rise in the incidence of syphilis infections is expected to contribute to the market growth in this country.
- Additionally, the development of various advanced products in this region is also expected to drive the market's growth. For instance, in March 2022, MedMira Inc launched the clinical trials of its product Reveal TP in Canada. Reveal TP is a point-of-care diagnostic for syphilis testing. Furthermore, technological advancements and the launch of new products, high healthcare spending, and the presence of well-established healthcare infrastructure are also fueling the overall regional market's growth to a large extent.
Rapid Plasma Reagin Test Industry Overview
The rapid plasma reagin test market is moderately fragmented and consists of several major players. In terms of market share, a few of the major players are currently dominating the market. Some of the key players in the market are Cardinal Health Inc., Thermo Fisher Scientific, Abbott Laboratories (Alere), F. Hoffmann-La Roche Ltd, Danaher Corporation (Beckman Coulter Inc.), and Siemens Healthcare, among others.
Rapid Plasma Reagin Test Market Leaders
-
Cardinal Health, Inc
-
Thermo Fisher Scientific
-
Abbott Laboratories (Alere)
-
F. Hoffmann-La Roche Ltd
-
Bio-Rad Laboratories
*Disclaimer: Major Players sorted in no particular order
Rapid Plasma Reagin Test Market News
- July 2022: Arlington Scientific Inc introduced an intelligent rack-loading system for the ASI Evolution Automated PRP syphilis analyzer. The ASI Smart Rack is an intelligent rack system that makes loading and scanning, and scanning sample tubes quicker and easier.
- March 2022: MedMira launched the Reveal TP product, an rapid plasma reagin test in Europe, and gets clinical trials underway in Canada. The company received the CE mark and is available in Europe and any country accepting the CE mark.
Rapid Plasma Reagin Test Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Increasing Incidence of Syphilis
4.2.2 Technological Advancement in Rapid Plasma Reagin Testing
4.2.3 Growing Awareness Regarding the Morbidity of Sexually Transmitted Diseases
4.3 Market Restraints
4.3.1 Lack of Adoption Regarding Disease Screening in Developing Countries
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
5.1 By Product Type
5.1.1 Rapid Plasma Reagin Test Kits
5.1.2 Analyzers and Instruments
5.1.3 Reagents
5.2 By End-User
5.2.1 Hospitals
5.2.2 Diagnostic Laboratories
5.2.3 Other End-Users
5.3 Geography
5.3.1 North America
5.3.1.1 United States
5.3.1.2 Canada
5.3.1.3 Mexico
5.3.2 Europe
5.3.2.1 Germany
5.3.2.2 United Kingdom
5.3.2.3 France
5.3.2.4 Italy
5.3.2.5 Spain
5.3.2.6 Rest of Europe
5.3.3 Asia-Pacific
5.3.3.1 China
5.3.3.2 Japan
5.3.3.3 India
5.3.3.4 Australia
5.3.3.5 South Korea
5.3.3.6 Rest of Asia-Pacific
5.3.4 Middle East and Africa
5.3.4.1 GCC
5.3.4.2 South Africa
5.3.4.3 Rest of Middle East and Africa
5.3.5 South America
5.3.5.1 Brazil
5.3.5.2 Argentina
5.3.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Cardinal Health Inc.
6.1.2 Thermo Fisher Scientific
6.1.3 Abbott Laboratories (Alere)
6.1.4 F. Hoffmann-La Roche Ltd
6.1.5 Bio-Rad Laboratories
6.1.6 Siemens Healthcare
6.1.7 AccuBioTech Co. Ltd
6.1.8 Becton Dickinson and Company
6.1.9 Danaher Corporation (Beckman Coulter Inc.)
6.1.10 bioLytical Laboratories Inc.
6.1.11 Sekisui Diagnostics
6.1.12 Novacyt Group
6.1.13 Sclavo Diagnostics
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Rapid Plasma Reagin Test Industry Segmentation
As per the scope of the report, the rapid plasma reagin (RPR) test is a serological test that is used to screen for syphilis, a sexually transmitted infection caused by the spirochete bacterium treponema pallidum.
The rapid plasma reagin test market is segmented by product type (rapid plasma reagin test kits, analyzers and instruments, and reagents), end user (hospitals, diagnostic laboratories, and other end users), and geography (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The report also covers the estimated market sizes and trends for 17 countries across major regions globally.
The report offers the value (in USD million) for the above segments.
| By Product Type | |
| Rapid Plasma Reagin Test Kits | |
| Analyzers and Instruments | |
| Reagents |
| By End-User | |
| Hospitals | |
| Diagnostic Laboratories | |
| Other End-Users |
| Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Rapid Plasma Reagin Test Market Research FAQs
What is the current Rapid Plasma Reagin Test Market size?
The Rapid Plasma Reagin Test Market is projected to register a CAGR of 4.10% during the forecast period (2024-2029)
Who are the key players in Rapid Plasma Reagin Test Market?
Cardinal Health, Inc, Thermo Fisher Scientific, Abbott Laboratories (Alere), F. Hoffmann-La Roche Ltd and Bio-Rad Laboratories are the major companies operating in the Rapid Plasma Reagin Test Market.
Which is the fastest growing region in Rapid Plasma Reagin Test Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Rapid Plasma Reagin Test Market?
In 2024, the North America accounts for the largest market share in Rapid Plasma Reagin Test Market.
What years does this Rapid Plasma Reagin Test Market cover?
The report covers the Rapid Plasma Reagin Test Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Rapid Plasma Reagin Test Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Rapid Plasma Reagin Test Industry Report
Statistics for the 2024 Rapid Plasma Reagin Test market share, size and revenue growth rate, created by ����vlog��ý™ Industry Reports. Rapid Plasma Reagin Test analysis includes a market forecast outlook to for 2024 to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.



